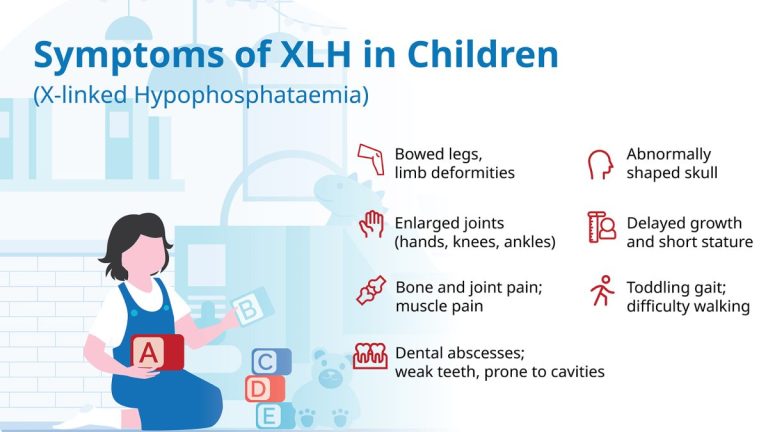
Thai Society for Pediatric Endocrinology and DKSH Raise Awareness of Rare Genetic Bone Disorder XLH
The Thai Society for Pediatric Endocrinology, DKSH, the Thai Rare Disease Foundation, and the XLH Community collaborate to raise awareness of the rare genetic bone disorder X-linked hypophosphatemia,…